SHANGHAI, China, Feb. 25, 2026 (GLOBE NEWSWIRE) -- MindRank, a clinical-stage artificial intelligence (AI)-empowered drug discovery company, today announced that the first patient has been dosed in the Phase III clinical trial of MDR-001, its AI-designed oral small-molecule GLP-1 receptor agonist (GLP-1RA) for the treatment of obesity and type 2 diabetes in China.
The Phase III study, MOBILE, is a multicenter, randomized, double-blind, placebo-controlled trial evaluating the long-term efficacy and safety of MDR-001 in approximately 750 patients across 50 clinical centers in China. The study is led by Professor Linong Ji of Peking University People’s Hospital.
MDR-001 is a biased, selective oral GLP-1 receptor agonist discovered and optimized using MindRank’s proprietary AI-enabled drug discovery platform. In a completed Phase IIb trial, MDR-001 demonstrated clinically meaningful efficacy, favorable tolerability, and improvements in cardiometabolic parameters, supporting its potential as a best-in-class therapy.
The molecule was identified using MindRank’s proprietary AI platform - Molecule Pro™, which is an end-to-end drug discovery platform to facilitate lead optimization across critical parameters. The program progressed from project initiation to U.S. IND clearance in 19 months and advanced to Phase III within approximately 4.5 years.
“Dosing the first patient in Phase III represents a major milestone for MDR-001 and for AI-enabled drug development,” said Zhangming Niu, Founder and Chief Executive Officer of MindRank. “We believe MDR-001 demonstrates how AI-driven discovery can accelerate innovation and deliver differentiated therapies to patients.”
About MDR-001
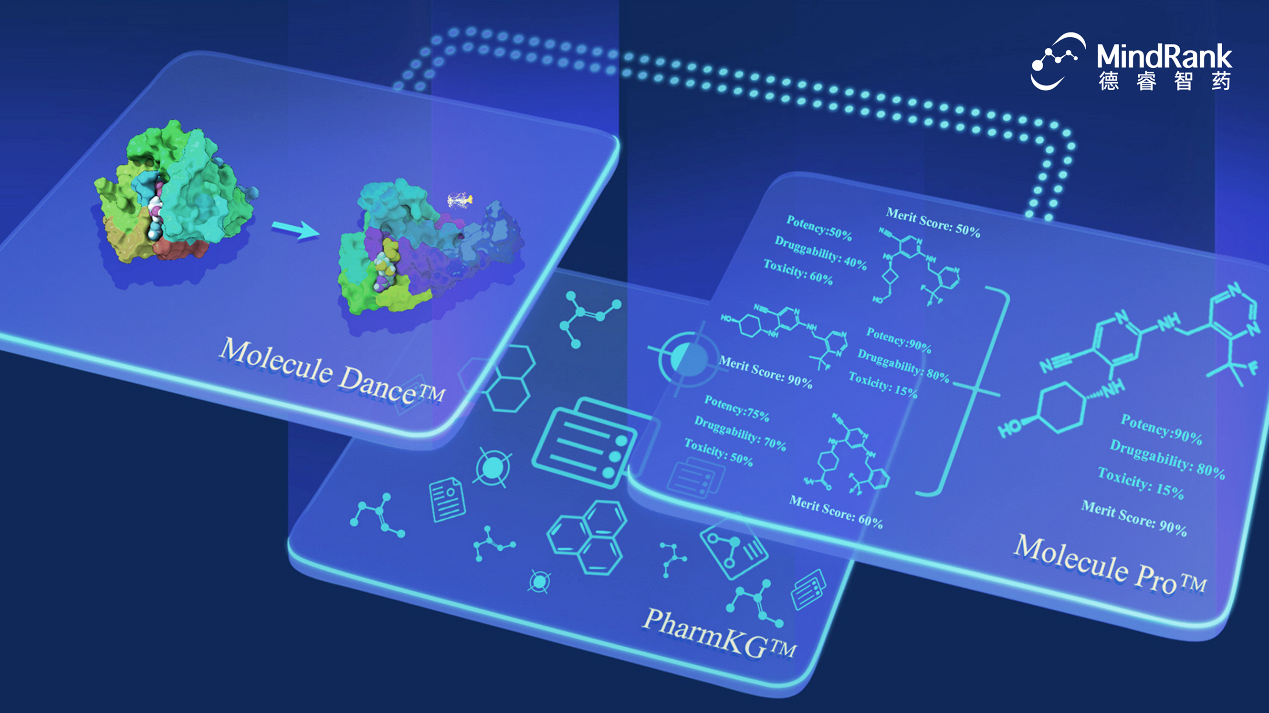
MDR-001 was developed by MindRank’s expert team utilizing the PharmKG™ biological knowledge graph to integrate vast amounts of literature and biological pathway analysis. By leveraging Molecule Dance™ for protein dynamic simulation modeling and biological validation iterations, the team deeply analyzed the GPCR protein-small molecule binding conformation and signal transduction mechanisms. This allowed for the quantification of specific biological pathway activities,including cAMP agonism and biased β-arrestin 2 recruitment,guiding the design of a highly selective GLP-1RA small molecule. The Molecule Pro™ platform’s generative AI and ADMET prediction modules, combined with wet-lab iterative feedback, enabled systematic prediction and optimization of the candidate’s structural druggability and safety.
About Molecule Pro™
Molecule Pro™ is MindRank’s proprietary AI-driven drug discovery platform. It deeply integrates expertise in artificial intelligence, computational chemistry, chemistry, biology, and medicine, driven by deep learning, generative AI, and large language models. The platform integrates nearly one hundred original algorithmic models, with core modules such as Large Language Model Knowledge Graphs and ADMET prediction reaching international leading standards. Molecule Pro™ focuses on "undruggable" targets, utilizing multi-objective optimization strategies to synergistically balance activity, selectivity, pharmacokinetics, and safety. By introducing prediction mechanisms for toxicity and off-target risks to avoid groups capable of causing adverse reactions (such as structural fragments leading to liver injury), the platform precisely designs and optimizes molecules, systematically improving the efficiency and success rate of drug development.
About MindRank
MindRank is an AI-driven biotechnology company dedicated to accelerating the implementation of "AI for Science" in drug discovery and leading paradigm shifts. Relying on an AI platform that deeply integrates biology, structural biology, chemistry, and medicine, the company focuses on tackling "hard-to-drug" targets to improve the efficiency and success rate of new drug development, aiming to provide global patients with innovative therapies offering greater clinical benefits.
For more information, visit www.mindrank.ai.
For BD inquiries, contact bd@mindrank.ai.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/cfc6fd9b-2fe6-4a41-8faa-7462868f198b

